 Their atoms are smaller than those in Group 1. Group 2 includes the Alkaline Earth Metals like calcium and magnesium, which bond easily. Some of them even explode when they come into contact with water or air. Group 1 consists of Alkali Metals - things like sodium, potassium,and cesium, which are not as dense as other metals and are highly reactive. Valence electrons are the number of electrons an element has available to bond with other elements to make molecules. This is the number of electrons an element has in its outer shell, which are known as valence electrons. A Roman numeral - or, in the modern periodic table, an Arabic numeral - designates each column. Groups (Vertical Columns)Įlements are grouped into columns based on their chemical properties. This image color-codes the different categories of elements. The periodic table is organized into groups and periods. How many elements can you guess just by looking at their atomic symbol? “Argentum” later became the French word for the same color - argent. 
Mercury has the designation Hg because of its Greek name, hydrargyrum, which means “liquid silver.” Silver, meanwhile, has the designation Ag for its Latin name, argentum. Potassium has the designation K because of its Neo-Latin name, kalium. In others - like potassium or mercury - scientists have to look back into history. In most cases - like oxygen or carbon - it’s simply the first letter of the element’s name. Atomic Symbolįinally, an element’s name determines the atom’s symbol. Future research can change the listed atomic weight of these elements.Īs you read the periodic table from left to right, atomic mass tends to increase. The atomic weight of these elements is based on the element that survived longest when it was created. These artificial elements don’t exist naturally and haven’t survived longer than several milliseconds at a time. Scientists created elements 93 through 118 in a laboratory. So, beryllium has four protons and five neutrons. Nine total protons and neutrons minus four protons leaves you with five neutrons. Beryllium is number four on the periodic table, so it has four protons. 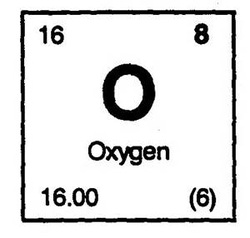
Remember that the atomic number tells you how many protons there are. That’s the total number of neutrons and protons combined in the nucleus of a beryllium atom.įrom there, you can find the number of neutrons in an atom by subtracting the total number of protons from the mass number. For example, beryllium’s atomic mass is 9.0112, so you can round it to nine. If you simply want to know an element’s mass number - which doesn’t have a decimal, because it’s the actual number of protons and neutrons in an atom - round the atomic mass to the nearest whole number. That’s why many atomic mass numbers have a decimal. In other words, the number you’re seeing is the element’s average mass, but you might find it at a different mass depending on the number of neutrons it contains. The atomic mass number on the modern periodic table is the weighted average of the atomic masses of that element’s naturally occurring isotopes. Therefore, different isotopes will have different amounts of mass. Just as an ion is an element with more or fewer electrons than usual, an isotope is a version of an element that contains a different number of neutrons. That’s because they sometimes contain different numbers of neutrons. However, even two atoms of the same element might have different weights. If you read the periodic table, you’ll see that oxygen weighs 15.999 u. This number tells you how much a single atom of that element weighs in atomic mass units. The atomic mass is the number usually - but not always - located at the bottom of an element key. That just tells you the number of protons. Therefore, don’t assume how many electrons an element has based on its atomic number on the periodic table. Cations are positively charged, while anions have a negative charge. These positively and negatively charged atoms are called ions. Others have fewer electrons than protons, giving them a positive charge. Some atoms have more electrons than protons, giving them a negative charge. However, this is only sometimes the case. It’s worth noting that most atoms have an equal number of protons and electrons, so the atomic number gives you a hint as to how many electrons an atom contains. Each element has one more proton than the one before it. Not only does it let you count the protons, but it also determines the element’s place on the table. The number of protons in an element cannot change or else it would become a different element entirely!Įvery element on the periodic table has an atomic number assigned to it. Since oxygen is number 8 on the periodic table, that tells you there are eight positive protons in the center of each atom. Atomic NumberĪn element’s atomic number lets you know how many protons are in the nucleus. The element key helps you learn about the individual elements in the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
